ARC Newsletter:
COVID-19
Below are all Antibiotic Resistance Coalition (ARC) Newsletter items classified under the topic area COVID-19. The search button below can be used to search across the page and all articles are listed in reverse chronological order.
March 2021
WHO details the impact of COVID-19 on tuberculosis in 2020
On March 24, 2021, marking World TB Day, the World Health Organization released data on the impact of COVID-19 on tuberculosis detection and mortality through 2020. According to this data based on 84 countries across the world, an estimated 1.4 million fewer people were able to receive care for tuberculosis in 2020 compared to 2019 numbers, resulting in more than half a million deaths. This reduction in care represented a more than 20% decrease from 2019, and among the 10 highest-burden countries, the overall reduction in tuberculosis diagnosis and care was estimated to be nearly 30%. In addressing tuberculosis, one of the world’s top infectious killers, WHO outlined several key recommendations, including scaling up simultaneous testing for both COVID-19 and tuberculosis and building civil society engagement to narrow gaps in tuberculosis and/or COVID-19 care, among others. The full World TB Day campaign page can be found here.
More than half of COVID-19 patients found to receive antibiotics
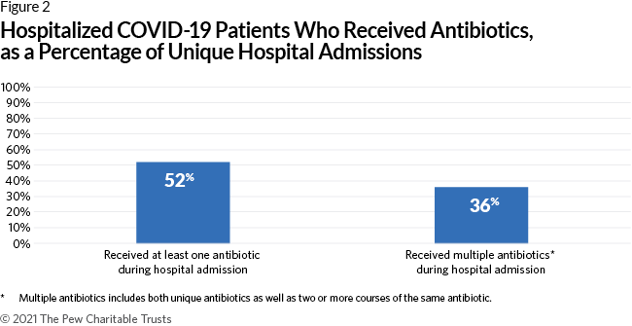
Using data from IBM Watson Health’s electronic health records database, Pew Charitable Trust analyzed antibiotic use in hospitalized COVID-19 patients. Looking at data from 5,000 U.S. patients from February through July 2020, more than half of COVID-19 hospital admissions resulted in one or more antibiotics being prescribed. While antibiotic prescribing was high, only a fifth of patients were diagnosed with suspected or confirmed bacterial pneumonia. These findings highlight the important role of institutional antimicrobial stewardship programs. The report states that ASPs should have the “resources necessary to ensure physicians … make the best antibiotic treatment decisions.”
No evidence of azithromycin in reducing time to recovery, death due to COVID-19
Azithromycin used to treat older COVID-19 outpatients made no significant differences in time to recovery, death, or hospitalization due to COVID-19. Participants in the PRINCIPLE trial were those suspected to have COVID-19 for 14 days or fewer with a higher risk of severe disease due to age and/or comorbidities. The study included 500 participants who were randomized to receive azithromycin and standard care and 823 participants who were randomized to receive standard of care alone. Comparing the results of the two groups showed no statistically significant differences in the primary outcomes of time to recovery (a self-reported outcome), death, or hospitalization. No differences in these outcomes were observed when comparing the two groups on the basis of age, existence of comorbidities, duration of illness, symptom severity, or positive versus negative SARS-CoV-2 test results. The results of this study were consistent with other randomized controlled trials in the inpatient setting which showed no clinical benefit of adding azithromycin to standard of care for COVID-19 patients. Further, the addition of such therapy to existing regimens could contribute to antimicrobial resistance in the absence of bacterial co-infections.
February 2021
Azithromycin demonstrates no clinical benefits when added to standard of care for COVID-19 patients in UK study
The results of one arm of the Randomised Evaluation of COVID-19 Therapy (RECOVERY) clinical trial across 176 hospitals in the UK has shown that the addition of azithromycin to standard of care for treating moderate to severe COVID-19 patients has no significant benefits when examining survival, duration of hospital stay, hospital discharge, or invasive mechanical ventilation. The investigators of this study included azithromycin as a treatment in part due to its immunomodulatory effects of dampening inflammation and potential antiviral activity. In this study, 2,582 patients were randomized to receive azithromycin orally or intravenously for ten days (median duration of treatment of six days) in addition to standard of care for COVID-19 while 5,181 patients received standard of care alone. This is the fourth published study examining the efficacy of azithromycin for the treatment of hospitalized COVID-19 patients, the only one to examine the effects of azithromycin not in combination with another experimental therapy, and the largest of the four with regards to sample size. The primary outcome of 28-day mortality was 22% for both the azithromycin and standard of care patients. Patients receiving azithromycin and standard of care versus standard of care alone had similar outcomes for all secondary and subsidiary clinical outcomes including time to discharge (10 and 11 days, respectively), proportion of patients discharged (69% and 68%), and receiving invasive mechanical ventilation or dying (25% and 26%). The results of this study held across several demographic and clinical characteristics including age, ethnic, sex, duration of symptoms, level of respiratory support, and corticosteroid use. The authors concluded the findings of the study by advising against the use of azithromycin in COVID-19 patients without confirmed secondary bacterial infections to mitigate the risk of exacerbating drug resistance to this important antibiotic.
January 2021
New 2021 Access to Medicine Index shows widespread industry interest in COVID-19 while other emerging infectious diseases are left behind
The recently published 2021 Access to Medicine Index builds off the Access to Medicine Foundation’s work to rank 20 of the world’s largest pharmaceutical companies in governance, research and development (R&D), and product delivery as these areas relate to access for patients in low- and middle-income countries (LMICs). While the Index has covered industry activity on emerging infectious diseases including coronaviruses (e.g., SARS-CoV-1 and MERS-CoV) spanning back to 2016, industry interest in coronavirus has predictably surged, with 63 reported projects in the pipeline for COVID-19 from 17 out of 20 companies compared to no projects for coronaviruses in the previous Index. Fewer than a third (7/24) of COVID-19 projects in Phase II of clinical development or later were determined to have conditions in place (such as licensing agreements or pricing arrangements) to ensure broader access to patients in LMICs by June 2020. The Index, which covered activity between June 1, 2018 and May 31, 2020, captures a focused glimpse of the pandemic in its early stages, tracking the industry’s response following identification of the virus in early 2020 and the WHO’s declaration of COVID-19 as a pandemic in mid-March. Despite the attention from 85% of the assessed companies on COVID-19, no companies reported R&D activities for other key emerging infectious diseases identified by the WHO as posing “the greatest public health risk due to their epidemic potential and/or whether there is no or insufficient countermeasures” including Lassa fever, MERS-CoV, SARS, Nipah virus, and Rift Valley fever.
Beyond monitoring of infectious disease research, the Index also reports on activities in developing new antimicrobials and how such antimicrobials are made available to patients in LMICs following market approval. Noting the risk of a pandemic caused by antimicrobial-resistant infections, the Index reports that twelve companies had 34 R&D projects for antibiotics in their pipelines, targeting eight types of bacterial infections. Eight companies were reported to be developing 27 vaccine projects with implications for the prevention of antibiotic-resistant infections. Although projects in development and launched products for bacterial infections and emerging infectious diseases are followed by the Index, the level of granularity reported does not allow for comprehensive scrutiny of those projects and products without strong plans or strategies for access, nor does it report where projects have stalled or been dropped from the pipeline. Instead, examples and opportunities to improve access are given ad hoc in company report cards, such as a need for access plans for GSK’s Shigella vaccine candidates, MSD’s tedizolid (Sivextro), and Daiichi Sankyo’s multidrug-resistant tuberculosis diagnostic tests (Genoscholar); and for equitable pricing strategies and registration filings for Pfizer’s ceftazidime/avibactam (Zavicefta) and ceftaroline (Teflaro/Zinforo) in countries with high burdens of lower respiratory tract infections.
Antibiotic prescribing trends in the U.S. during the COVID-19 pandemic
A team of researchers in the U.S. used patient data from the IQVIA Total Patient Tracker to examine trends in outpatient antibiotic prescriptions between January and May 2020. The researchers found that 10.4 million fewer patients were dispensed antibiotics during this time period than expected, according to past prescribing trends. The largest decreases in antibiotic prescriptions were found to be among children, especially those below the age of four, as well as antibiotics used for surgical prophylaxis, dentistry, and respiratory infections.
Another review found that nearly three-fourths of all patients with COVID-19 received antibiotics between the years January and June 2020. However, less than a tenth (8.6%) of patients were estimated to have a bacterial coinfection. Prescribing prevalence was found to be higher as age increased, and increased antibiotic prescribing rates were also associated with increased proportions of patients requiring mechanical ventilation. These findings suggest that, despite decreased overall antibiotic prescribing trends, antibiotics are still used commonly among COVID-19 infected populations.
Impacts of increased hand-washing and facial coverings on the incidence of hospital-acquired infections
As the COVID-19 pandemic has resulted in behavior changes across the globe, researchers in Taiwan examined the impact of increased hand-washing, face masks, and other infection control measures in a 1700-bed Taiwanese medical center. Between January and May 2020, it was found that there was no significant decrease in overall incidence density of hospital-acquired infections. However, the total number of patients isolated with multidrug resistant infections was found to be significantly lower in 2020, compared to previous years. And when stratified by pathogen type, both carbapenem-resistant Acinetobacter baumannii (CRAB) and vancomycin-resistant Enterococcus (VRE) infections decreased significantly in 2020 when compared to previous years, along with slight decreases in both methicillin-resistant Staphylococcus aureus (MRSA) and carbapenem-resistant Pseudomonas aeruginosa (CRPA). These findings contrast with previous studies. During the 2003 SARS outbreak, increased use of gowns and gloves were found paradoxically to result in greater cross-transmission of MRSA infections in intensive care unit settings, potentially due to low compliance resulting in direct cross-transmission. However, the findings of this new study suggest a collateral benefit of COVID-19 infection prevention and control measures specifically on the number of patients isolated with multidrug resistant infections.